Weekly Dose of #73
Nuclear AI, Approved CRISPR, Diabetes Implants, Morning Sickness, MDMA Application, FunSearch
Hi friends 👋,
Happy Friday and welcome back to our 73rd Weekly Dose of Optimism. This is the most jam packed Weekly Dose in a long time. We thought we had the draft done, and then the good guys kept doing good things.
No circling back in the New Year just yet. The optimists are closing out 2023 strong and we’re here to cover it.
Let’s get to it.
Merge is a single API to add hundreds of integrations to your app. It’s loved and used by companies like Ramp, Navan, and hundreds of other tech companies. Here’s why:
Seamlessly integrate: hundreds of software products. Merge ensures a no hassle onboarding experience for your customers, secure data, and continuity across your software stack.
More, with less: Your budgets are lower, but your engineers still need to achieve more. With Merge, engineers can more quickly build and maintain all of the integrations your business needs to grow.
New Standard: Merge covers hundreds of integrations, handles the lifetime of maintenance, is fully secure and compliant, and let’s your team move faster.
Stop wasting your engineering team’s time building out dozens of bespoke, complex integrations and just use Merge – it’ll save you time and money. And here’s the kicker, Merge is offering Not Boring readers a $5,000 credit on annual plans.
(1) Microsoft Targets Nuclear to Power AI Operations
Jennifer Hiller for The Wall Street Journal
The generative AI experiments tap in to Microsoft’s investment in OpenAI, creator of the viral chatbot ChatGPT. For the past six months, a team of Microsoft employees have been training a large language model with U.S. nuclear regulatory and licensing documents, hoping to expedite the paperwork required for such approvals, which can take years and cost hundreds of millions of dollars.
When your partners need to train a special LLM to complete your permitting paperwork, you may have put just a bit too much regulatory red tape in place. But that’s exactly what Microsoft is doing to expedite its way through the NRC’s nuclear permitting process. It sounds like a very one-off, narrow task to train an AI for, but those time and cost savings are potentially huge.
The last nuclear operator to get approval from the NRC, NuScale, submitted a 12,000 page application and spent over $500M in the processes. Despite NuScale’s approval, the projected eventually collapsed because demand for the utility never hit critical mass. So the company spent years and over $500M on getting approved for what exactly? Not exactly an attractive proposition for the next nuclear upstart thinking of making a go at it. That’s why cutting the time and cost of the regulatory approval process is important; it lowers the hurdles for new entrants. (Plus, Microsoft brings its own demand.)
Microsoft is partnering with a nuclear advocacy organization to train an AI that could cut the human hours involved in the regulatory process by 90% — potentially saving years and millions of dollars in the process. To do so, they’re training their model on a very specific and structured set of regulatory filings, in order to be to basically produce identical copies in the future.
If Microsoft is actually able to shave time off the regulatory approval process, this could be as big a breakthrough as any new reactor design. Fighting regulation with technology, you absolutely love to see it.
In more good nuclear news, the Diablo Canyon nuclear plant in California, which was scheduled to be shut down, then kept alive temporarily, is getting extended beyond its scheduled 2025 closing date.
In even MORE good nuclear news, Kairos Energy received approval from the Nuclear Regulatory Commission to start construction on its Hermes reactor. This is massive because it’s the first construction permit application ever issued for a Gen-IV, or advanced, reactor. This is just a test reactor and will be built at Oak Ridge National Labs, but it’s an important step in the march to bring advanced reactors online.
(2) FDA Approves First Gene Therapies to Treat Patients with Sickle Cell Disease
From The FDA
Casgevy, a cell-based gene therapy, is approved for the treatment of sickle cell disease in patients 12 years of age and older with recurrent vaso-occlusive crises. Casgevy is the first FDA-approved therapy utilizing CRISPR/Cas9, a type of genome editing technology. Patients’ hematopoietic (blood) stem cells are modified by genome editing using CRISPR/Cas9 technology.
A couple of weeks ago, we covered the UK’s approval of the first CRISPR/Cas9 gene-edited therapy, CASGEVY.
Now, the FDA is following suit. It has approved two groundbreaking cell-based gene therapies, Casgevy and a new one, Lyfgenia, for treating sickle cell disease (SCD) in patients aged 12 and older. Notably, Casgevy is the first FDA-approved therapy utilizing CRISPR. Both treatments modify patients' blood stem cells to increase the production of types of hemoglobin that reduce the sickling of red blood cells, addressing the primary problem in SCD.
The use of CRISPR in Casgevy represents a major breakthrough in genome editing technology that could impact the treatment of any number of genetic disorders. CRISPR allows for precise modifications in DNA, so doctors can alter or correct the specific gene mutations that are causing diseases. It turns the treatment of diseases from a science problem into a coding problem. The therapeutic potential of CRISPR has been known for years now, but Casgevy approval is a big step towards putting that potential into practice.
The UK approval was massive, and the FDA approval may be even more important as it often has significant influence over global standards. The Century of Biology is well underway!
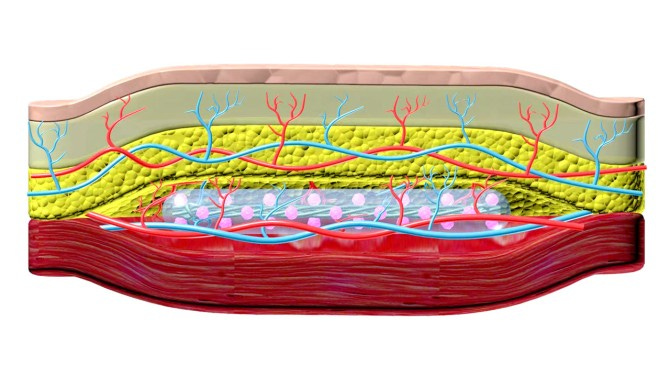
(3) Under-the-skin implant could treat Type I diabetes
David Nutt for Cornell Chronicle h/t Tim Sullivan
A collaboration between researchers from Cornell and University of Alberta, Edmonton, has created a new technique to treat Type 1 diabetes: implanting a device inside a pocket under the skin that can secrete insulin while avoiding the immunosuppression that typically stymies management of the disease. The approach would offer an easier, long-term and less invasive alternative to insulin injections or traditional transplants that require immunosuppression.
Researchers from Cornell and University of Alberta have developed a new technique for managing Type 1 diabetes. This technique involves implanting a thread-like device under the skin, which can secrete insulin and receive essential nutrients and oxygen, without requiring immunosuppression. This approach aims to offer a simpler, less invasive, and more sustainable alternative to insulin injections or traditional transplants. The researchers proved the effectiveness of the device in diabetic mice and pig trials, and hopes to engage in human trials soon.
About 1.6 million folks in the U.S. and nearly 10 million worldwide have Type 1 diabetes, and an effective, less invasive treatment would be more than welcomed. GCMs, pumps, and hybrid devices have gotten a lot better over the years, but an even less invasive mechanism for treatment could go a long way in reducing daily suffering.
(4) Extreme morning sickness? Scientists finally pinpoint a possible cause
Carissa Wong for Nature
Researchers have pinpointed a hormone released by growing fetuses that might cause a debilitating form of morning sickness. Women who are more sensitive to the hormone, which increases during early pregnancy, might be at greater risk of experiencing a severe form of nausea and vomiting, called hyperemesis gravidarum, according to their study.
OK, here’s a good one for our male readers with pregnant wives — the next time she’s experiencing morning sickness just explain to her that researchers recently discovered that the hormone GDF15 may cause severe morning sickness. And that women with higher sensitivity to this hormone, which increases during early pregnancy, are at greater risk of this severe morning sickness. I’m sure she’ll love to hear this from you and be very appreciative of your insights on knowledge!
The actual good news is that identifying the cause of morning sickness may lead to an eventual treatment, such as desensitizing women to GDF15 before pregnancy or using antibodies to block GDF15 or its receptors, to prevent or reduce severe nausea and vomiting during pregnancy.
(5) MAPS PBC Announces Submission of New Drug Application to the FDA for MDMA-Assisted Therapy for PTSD
From MAPS
MAPS Public Benefit Corporation, a clinical-stage company dedicated to changing the way mental health conditions are treated, announced the submission of a new drug application to the FDA for MDMA used in combination with psychological intervention, which includes psychotherapy, or talk therapy, and other supportive services provided by a qualified healthcare provider. This investigational MDMA-assisted therapy is in development for individuals with post-traumatic stress disorder and if approved, would be the first psychedelic-assisted therapy approved for PTSD.
This has been a long time coming and the folks behind the announcement have been fighting a particularly hard uphill battle — against stigma, big pharma, bad science, regulation, you name it — for decades to get to this point. MAPS is an organization that’s been around since 1986 leading research and galvanizing public support for the therapeutic use of psychedelics. This week it announced it submitted a new drug application to the FDA for an MDMA drug + psychological intervention protocol to be used in the treatment of PTSD. To the uninitiated, that sounds wild…the FDA is going to consider sanctioning Molly to be people suffering from PTSD?! But the thing is, MDMA when matched with proper interventions is ridiculously effective in treating PTSD (among other ailments.)
Take these Phase 3 clinical trial results published in Nature, for example, which found that 71.2% of participants in the MDMA-assisted therapy group no longer met the diagnostic criteria for PTSD by the end of the 18-week trial, compared to 47.6% in the placebo group. And that 67% of participants who received three MDMA-assisted therapy sessions no longer qualified for a PTSD diagnosis, with an 88% experiencing a clinically meaningful reduction in symptoms. This contrasts with 32% in the placebo group who no longer met the PTSD diagnosis and 60% experiencing a significant reduction in symptoms.
See what I mean? MDMA is highly effective at treating PTSD. Given that context, it’s crazy it’s taken the FDA so long to start considering it as a therapeutic.
Here’s to hoping MAPS’ application moves swiftly through the approval process and that everyone who needs these drugs get them as soon as possible.
(6) FunSearch: Making new discoveries in mathematical sciences using Large Language Models
From Google DeepMind
This work represents the first time a new discovery has been made for challenging open problems in science or mathematics using LLMs. FunSearch discovered new solutions for the cap set problem, a longstanding open problem in mathematics.
A sixth dose? I know, I know. But like we said, people just kept doing big stuff this week. Researchers at Google DeepMind paired a pre-trained LLM to come up with creative solutions and an automated evaluator to keep the LLM’s hallucinations and mistakes in check. The pair discovered new solutions to mathematical problems and more effective algorithms for “bin-packing.”
The authors wrote, “FunSearch demonstrates that if we safeguard against LLMs’ hallucinations, the power of these models can be harnessed not only to produce new mathematical discoveries, but also to reveal potentially impactful solutions to important real-world problems.”
DeepMind has been on a tear solving really hard problems, from AlphaFold to millions of new materials (just two weeks ago!) to generating new knowledge.
As gfodor said on X, “If DeepMind just definitively proved neural networks can generate genuinely new knowledge then it’s the most important discovery since fire.”
Bonus: Age of Miracles Episode 9: Fusion Founders
Packy here. We’re getting close to the end of our first season of Age of Miracles, and we’re heading into the finish with some momentum: we just crossed 100k listens on the season, and are dropping maybe the coolest episode we’ve done yet.
On it, we talk to founders, operators, and investors of startups working to make commercial fusion a reality in the next decade. We spoke with JC Btaiche at Fuse Energy, Ryan Umstattd and Derek Sutherland at Zap Energy, Francesco Sciortino at Proxima Fusion, and David Kirtley at Helion Energy, plus Clay Dumas and Dr. Clea Kolster at Lowercarbon Capital and Ian Hogarth at Plural Platforms to understand the different approaches companies are taking to putting fusion power on the grid.
Coming out of these interviews, I feel more bullish on fusion arriving sooner than anticipated than I have before. Now, it’s a question of how soon and who, not if.
We have geniuses among us tackling the world’s biggest problems. If you want a real double-shot of optimism, listen to these ones explain how they’re turning sci-fi into reality.
Listen on Spotify, Apple, or YouTube.
That’s all for this week. If you’re a developer and have some time this weekend, check out Merge.
We’ll be back in your inbox on Tuesday.
Thanks for reading,
Dan + Packy







Maybe the NRC will come up with an LLM to read the Microsoft generated regulatory filings and the computers can keep each other busy and allow us to finally build some reactors.
Lots of good news here. Thanks for putting this together.